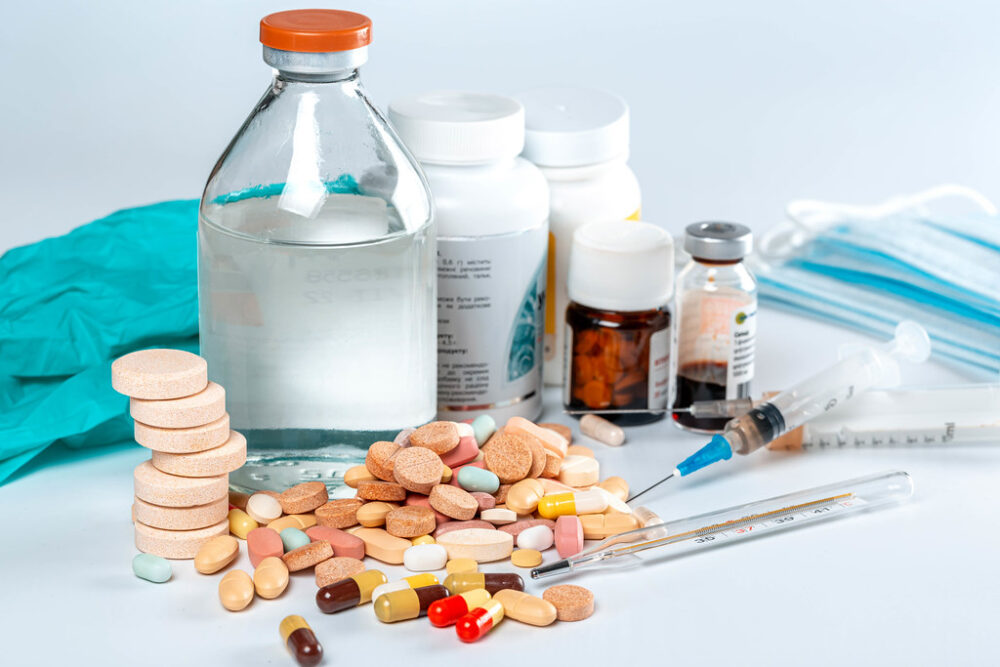
On Sept. 12, 2023, an FDA advisory committee concluded that over-the-counter (OTC) oral phenylephrine — the main active ingredient in Sudafed PE — is ineffective as a treatment for nasal congestion. This finding did not strike healthcare practitioners as “especially surprising” given that its efficacy has been controversial for decades. Despite this, the ingredient is in at least 250 different cough and cold products and remains a popular option for Americans during cold and flu season. In 2022, 242 million bottles (or packages) of products containing phenylephrine were sold, creating nearly $1.8 billion in sales.
The evidence presented at the two-day advisory committee meeting did not support the efficacy of oral phenylephrine for nasal decongestion compared to placebo. A placebo is a pharmacologically inert substance used as a control in testing new drugs, such as a sugar pill. A meta-analysis conducted in 2007 pushed the FDA to start reviewing efficacy data that year and was also among the studies presented at the 2023 advisory committee meeting. This study found that, based on data from eight studies that included 138 patients, there was no difference between oral phenylephrine and placebo on nasal airway resistance (NAR) in patients with nasal congestion.
“It is only the oral route that is ineffective because the drug must first pass the gastrointestinal tract and the liver — where it is rapidly metabolized, or broken down — before it reaches the nose.”
These findings led the study authors to submit an FDA citizen petition in 2007, requesting changes to the drug’s dosing on its monograph. An FDA citizen petition is a process that allows individuals and community organizations to ask the FDA to issue, amend, or revoke a regulation. Following four additional studies that also failed to demonstrate the efficacy of oral phenylephrine, the same study authors submitted an additional citizen petition in 2015 — this time requesting that oral phenylephrine be removed from the OTC monograph for nasal decongestant products entirely. An OTC drug monograph is a “rulebook” for each category of drug (e.g., nasal decongestants) that recognizes which active ingredients are allowed along with their approved uses, doses, routes of administration, labeling, and testing. If an OTC drug adheres to the requirements in a final FDA monograph, it can be manufactured and marketed in the United States without FDA pre-approval.
It is important to note that this data applies specifically to the oral formulation of phenylephrine; nasal sprays that contain the ingredient are still very effective. It is only the oral route that is ineffective because the drug must first pass the gastrointestinal tract and the liver — where it is rapidly metabolized, or broken down — before it reaches the nose. Thus, there is very little drug that actually reaches the nose to treat the congestion. While oral phenylephrine itself is not useful, it is not dangerous. The multi-product formulations it is often found in (such as Tylenol Cold & Flu Severe, Vicks DayQuil, and Mucinex Sinus-Max) may offer some relief through their other ingredients.
What popularized phenylephrine as a decongestant was the Combat Methamphetamine Epidemic Act of 2005 (CMEA), which was enacted to place restrictions on over-the-counter medications that could be used to produce illicit methamphetamines. In order to curb its abuse, the CMEA required that the OTC medication pseudoephedrine (Sudafed) — which has well-documented data for relieving nasal congestion — be moved “behind the counter” of U.S. pharmacies and mandated that purchasers present photo identification. In response to the CMEA and concern about sales, drug manufacturers quickly reformulated most of their OTC products to replace pseudoephedrine with phenylephrine. In other words, while Sudafed was moved behind the counter and became subject to purchasing restrictions, Sudafed PE was easily accessible on pharmacy shelves.
“While Sudafed was moved behind the counter and became subject to purchasing restrictions, Sudafed PE was easily accessible on pharmacy shelves.”
It is certainly in the best interest of the consumer to quickly remove placebo-like products from the market, as they only create unnecessary costs and delay sickness recovery. There are also significant ethical implications associated with drug manufacturers marketing an ineffective product to consumers. However, the events to follow the advisory committee’s decision are still unclear. Multiple class-action lawsuits against Johnson & Johnson, the manufacturer of name-brand Sudafed PE, have already been filed by citizens across multiple states. The FDA will now need to officially decide whether to remove the drug’s status as Generally Recognized as Safe and Effective (GRASE), which would lead to its removal from the monograph. This decision could potentially be delayed for months by lawsuits and lobbying instigated by drug manufacturers, which leaves the ultimate fate of phenylephrine still up in the air.